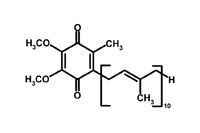
 Coenzyme Q10 (2,3 dimethoxy-5 methyl-6-decaprenyl benzoquinone) is a fat-soluble, quinone nutrient [1,2]. It is a ubiquitous compound necessary for a number of reactions related to energy metabolism and is vital for the proper transfer of electrons within the mitochondrial oxidative respiratory chain. Since dysfunctional energy metabolism has been implicated as a contributing factor for a number of conditions, coenzyme Q10 has been utilized in the treatment of cardiac, neurologic, and immunologic disorders. Coenzyme Q10 has also been demonstrated to prevent lipid peroxidation as an antioxidant [3].
Coenzyme Q10 (2,3 dimethoxy-5 methyl-6-decaprenyl benzoquinone) is a fat-soluble, quinone nutrient [1,2]. It is a ubiquitous compound necessary for a number of reactions related to energy metabolism and is vital for the proper transfer of electrons within the mitochondrial oxidative respiratory chain. Since dysfunctional energy metabolism has been implicated as a contributing factor for a number of conditions, coenzyme Q10 has been utilized in the treatment of cardiac, neurologic, and immunologic disorders. Coenzyme Q10 has also been demonstrated to prevent lipid peroxidation as an antioxidant [3].
Coenzyme Q10 and Neurologic Conditions
The effects of Coenzyme Q10 on neurologic conditions have been impressive. A randomized, multicenter study of eighty patients found that 1,200 mg per day of coenzyme Q10 was associated with up to 44% less functional decline in patients with Parkinson’s disease [4]. An additional study demonstrated mild symptom improvement with daily oral dosing of 360 mg of coenzyme Q10 [5]. Researchers have found that coenzyme Q10 can prevent and even stabilize mitochondrial abnormalities. Mitochondrial abnormalities have been demonstrated in several age-related disorders and are particularly prominent within both Alzheimer’s and Parkinson’s disease patients. In fact, main researcher Ikebe found seventeen times the level of mitochondrial DNA abnormalities in the striatum of Parkinson’s disease patients when compared with control subjects [6]. The positive effects Coenzyme Q10 has displayed in its use with Parkinson’s disease may be secondary to its ability to prevent or stabilize damaged mitochondria.
Elevated levels of cortical mitochondrial DNA deletions have also been found in patients with Alzheimer’s disease [7]. Currently, there are no published controlled clinical trials of Coenzyme Q10 in Alzheimer’s disease. Since Alzheimer’s disease is a neurodegenerative disease with known defect in the inner mitochondrial membrane, oral administration of CoQ10 may show benefit in patients with AD.
Coenzyme Q10 has also been found to be beneficial in the treatment of migraine headaches. A preliminary trial of 32 patients taking 150 mg of coenzyme Q10 daily reduced the frequency of migraine attacks [8]. A randomized double-blind, placebo-controlled trial of 42 patients taking coenzyme Q10 at 300 mg a day was also found to be beneficial [9].
Although the Dietary Supplement Health and Education Act of 1994 does not allow claims for treatment of specific diseases in the United States, coenzyme Q10 has been cleared for treatment indications in other countries, such as for congestive heart failure (CHF) in Japan since 1974.
- Greenberg S, Frishman WH. Co-enzyme Q10: a new drug for cardiovascular disease. J Clin Pharmacol 1990;30:596-608.
- Tran MT, Mitchell TM, Kennedy DT, Giles JT. Role of coenzyme Q10 in chronic heart failure, angina, and hypertension. Pharmacotherapy 2001;21:797-806.
- Sugiyama S, Kitazawa M, Ozawa T, Suzuki K, Izawa Y. Anti-oxidative effect of coenzyme Q10. Experentia 1980;36:1002-3.
- Shults CW, Oakes D, Kieburtz K, Beal MF, Haas R, Plumb S, et al. Effects of coenzyme Q10 in early Parkinson disease: evidence of slowing of the functional decline. Arch Neurol 2002;59:1541-50.
- Muller T, Buttner T, Gholipour AF, Kuhn W. Coenzyme Q10 supplementation provides mild symptomatic benefit in patients with Parkinson’s disease. Neurosci Lett 2003;341:201-4.
- Ikebe S. et al. Increase of deleted mitochondrial DNA in the striatum in Parkinson’s disease and senescence. Biochem Biophys Res Commun. 1990; 170: 1044.
- Corral-Debrinski M. et al. Marked changes in mitochondrial DNA deletion levels in Alzheimer brains. Genomics. 1994; 23: 471.
- Rozen TD, Oshinsky ML, Gebeline CA, Bradley KC, Young WB, Shechter AL, et al. Open label trial of coenzyme Q10 as a migraine preventive. Cephalalgia 2002;22:137-41.
- Sandor PS, Di Clemente L, Coppola G, Saenger U, Fumal A, Magis D, et al. Efficacy of coenzyme Q10 in migraine prophylaxis: a randomized controlled trial. Neurology 2005;64:713-5